UPNEEQ®
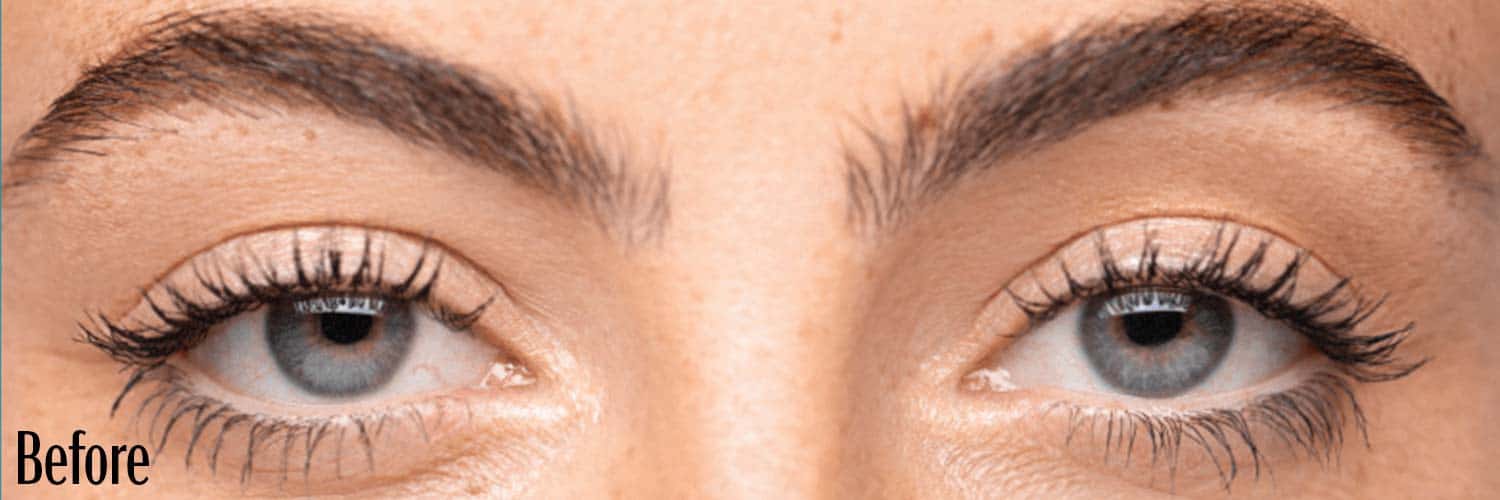
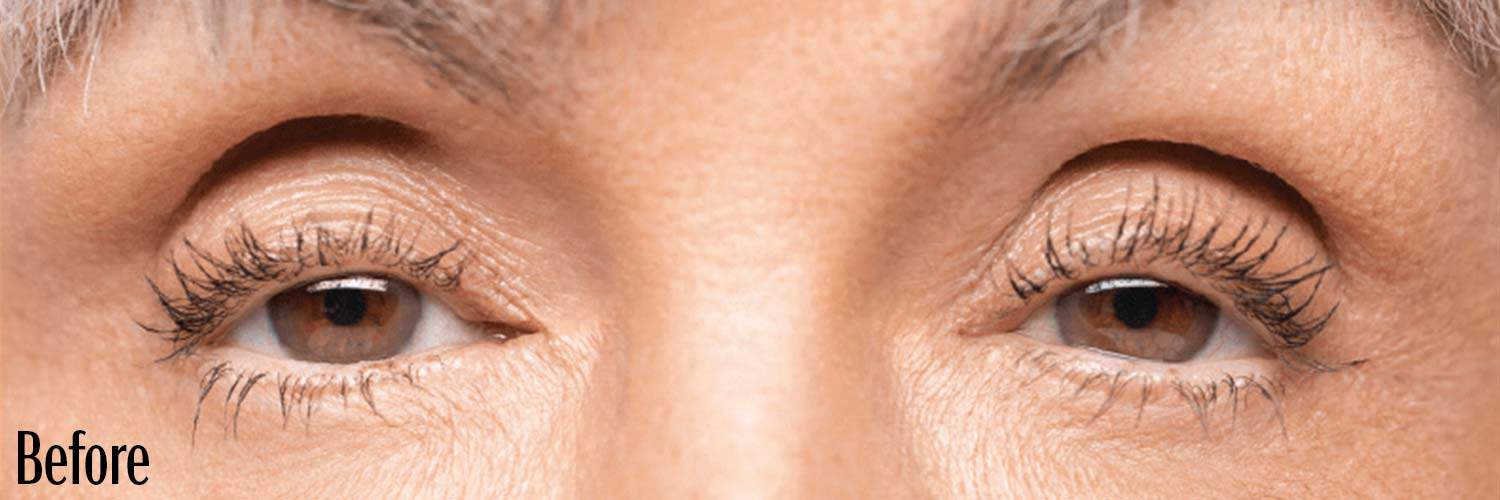
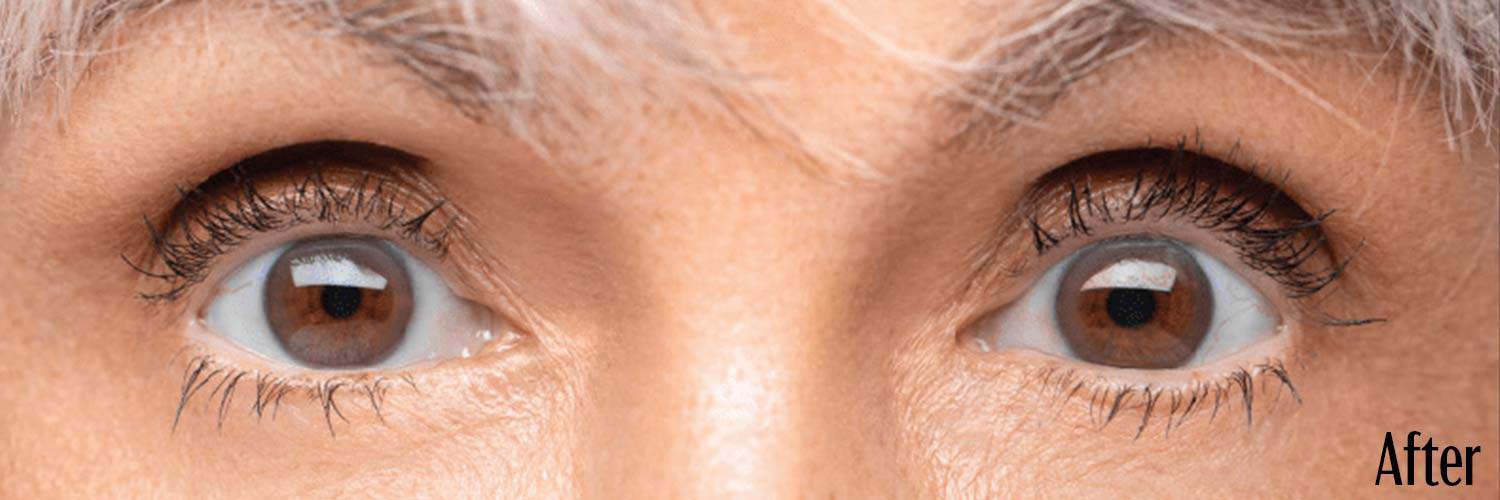
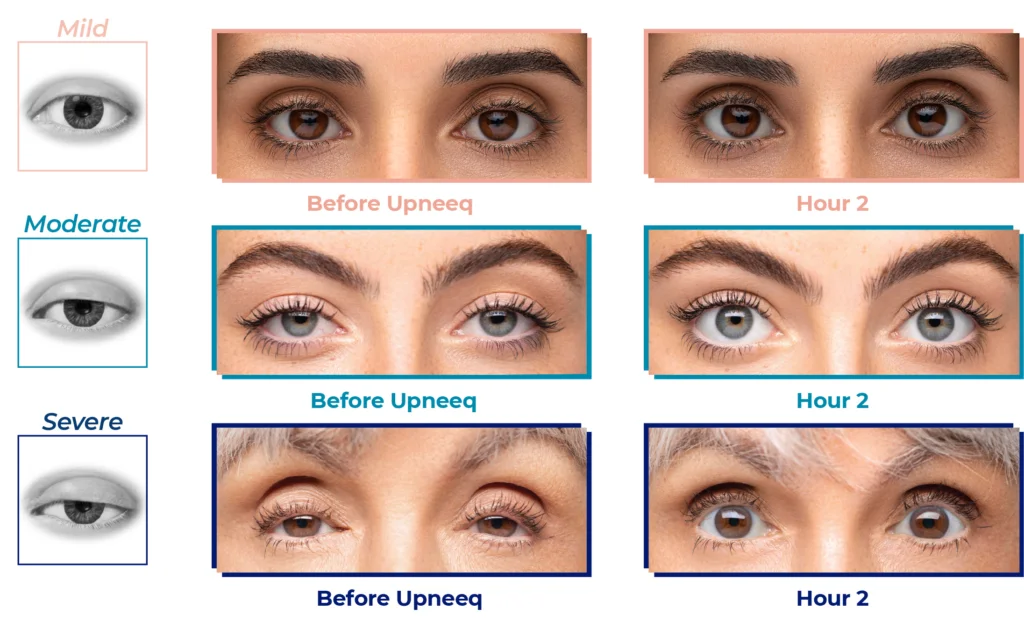
Before and After
WHY DO MY EYELIDS LOOK LOW?
If your eyes look “tired” or “sleepy,” you may have a condition called acquired blepharoptosis (also known as acquired ptosis or low-lying lids).
REQUEST A CONSULTATIONIMPACT OF LOW-LYING LIDS
Low-lying lids can affect the way you look and see things.
REQUEST A CONSULTATIONUPNEEQ®
Upneeq is the only FDA-approved prescription eyedrop for acquired ptosis (low-lying lids) that lifts your upper eyelids to open your eyes
Upneeq is the only FDA-approved prescription eyedrop for acquired ptosis (low-lying lids) that lifts your upper eyelids to open your eyes
UPNEEQ ®
(oxymetazoline hydrochloride ophthalmic solution), 0.1%
Low-lying lids may be caused by acquired blepharoptosis.
If you see signs of low-lying lids, talk to your RSVP Esthetician about getting UPLIFTED with Upneeq!
- PRODUCT INFORMATION
- Warnings And Precautions
- Adverse Reactions
- Drug Interactions
- Report Adverse Reactions
References: 1. Upneeq® (oxymetazoline hydrochloride ophthalmic solution) 0.1%. [Prescribing Information]. RVL Pharmaceuticals, Inc. 2020. 2. Finsterer J. Ptosis: causes, presentation, and management. Aesthetic Plast Surg. 2003;27(3):193-204. 3. Richards HS, Jenkinson E, Rumsey N,et al. The psychological well- being and appearance concerns of patients presenting with ptosis. Eye. 2014; 28(3):296-302. 4. Data on file, RVL Pharmaceuticals, Inc
Frequently Asked Questions
What is Upneeq?
How does Upneeq work?
How fast does it work?
How long do the results last?
How do I use Upneeq?
Can I use Upneeq with contact lenses?
Is Upneeq safe to use every day?
Who is a good candidate for Upneeq?
Who should avoid using Upneeq?
Are there any side effects?
Will Upneeq affect my vision?
Can I combine Upneeq with cosmetic treatments?
How is Upneeq different from surgery?
How should I store Upneeq?
Request a Consultation
Are you ready to take the first step to achieving your goals? Request a consultation today! Or, if you’re ready to book your appointment, click the button below and we will reach out to you.
Book appointment